  
Has to be heated strongly and so does the iron wool. Bromine displaces iodide ions from solution. The reaction is faster than that of iodine but slower than that of chlorine. Discussion: Chlorine displaces both bromide and iodide ions from solution. Has to be warmed and the iron wool heated. Reacts with heated iron wool very quickly, although not as quickly as fluorine does. For the word puzzle clue of what is the trend in reactivity down group 7, the Sporcle Puzzle Library found the following results. Very few scientists handle fluorine because it is so dangerous. You can see the trend in reactivity if you react the halogens with iron wool. Fluorine is the most reactive halogen because there is only one shell of electrons between the nucleus and outer shell, so its atoms gain an outer electron most. įluorine is the most reactive element of all in Group 7. This is the opposite trend to that seen in the alkali metals in Group 1 of the periodic table. The video below provides an overview of the halogens.The non-metal elements in Group 7 – known as the halogens – get less reactive as you go down the group. Halogen – One of the 5 non-metals in group 7 of the periodic table.ĭisplacement reaction – A reaction in which a more reactive element takes the place of a less reactive element in a compound. Halogens become less reactive as you go down group 7 in the periodic table, because the outer electron shell gets further away from the attraction of the nucleus, and so an electron is gained less easily.Ī less reactive halogen will be displaced by a more reactive one from an aqueous solution of its metal halide.Ĭhlorine + potassium bromide → potassium chloride + bromine Iodine (State of matter at room temperature) Solid (Colour) Grey/ black Reactivity series If you test different combinations of the halogens and their salts, you can work out a reactivity series for Group 7: the most reactive halogen displaces all of the other. They form hydrogen halides, which dissolve in water, forming acidic solutions.įluorine (State of matter at room temperature) Gas (Colour) YellowĬhlorine (State of matter at room temperature) Gas (Colour) Greenīromine (State of matter at room temperature) Liquid (Colour) Red/ orange 5.1 Atomic structure and the periodic table. They form molecular compounds with non-metals. Explain how the observed simple properties of Group 7 depend on the outer shell of electrons of the atoms and predict properties from given trends down the group. Which of the following statements is true about the trend down group 7 (halogens) with increase in atomic number The reactivity increases. 
They react with metals to form ionic compounds where the halide ion has a charge of -1. They consist of molecules made up of two atoms (diatomic molecules). Reactivity of group 7 non-metals decreases as you go down the group However, halogen atoms form negative ions when they gain an electron to obtain a full outer. 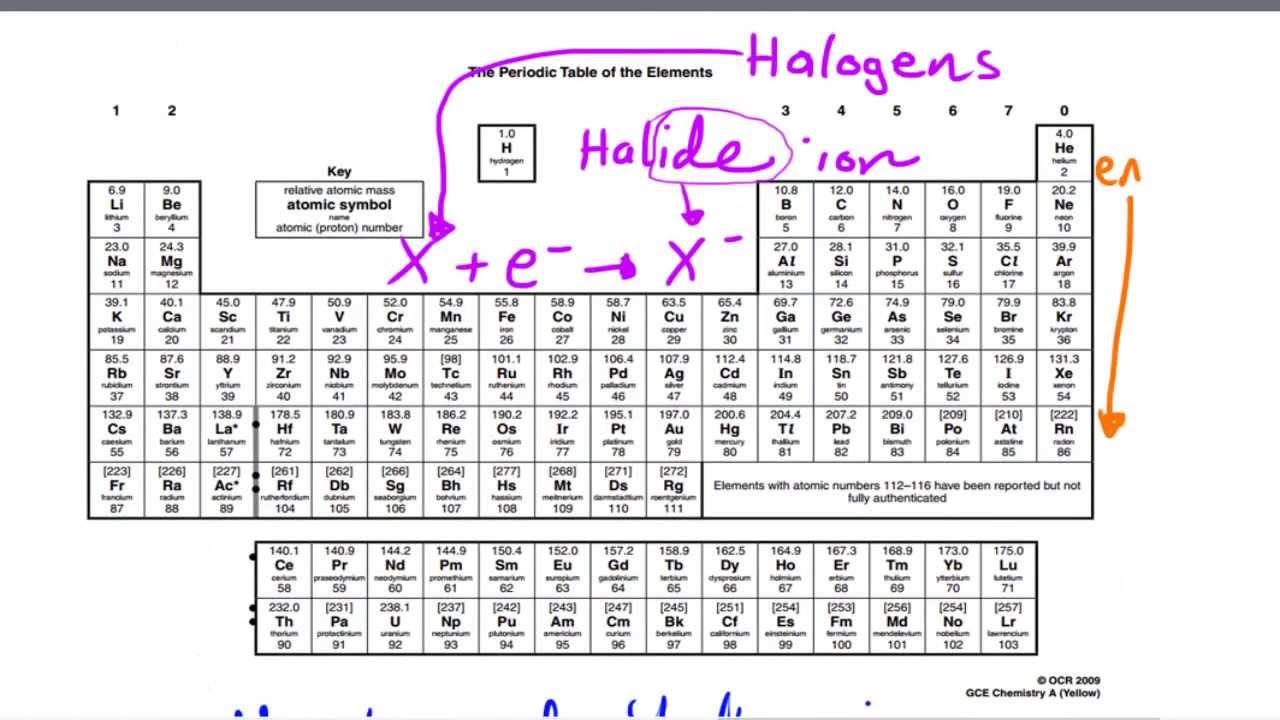
Two types of reactions might occur when concentrated sulfuric acid is added to a solid ionic halide like sodium fluoride, chloride, bromide or iodide. The halogens have the following properties: This page examines the redox reactions involving halide ions and concentrated sulfuric acid, using these reactions to discuss the trend in reducing ability of the ions from fluoride to iodide. Group 7 of the periodic table is home to the Halogens. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
